Wheres Austin when u need him
Wheres Austin when u need him
Being a chemist, I've never figured out where the loss in mass amount from... It's just not my field and even physical chemists don't really bother themselves with nuclear physics.Originally posted by Big RICHARD
The reason I asked is because energy is effectively created from mass.
Back to your point, you are completely correct in quoting the equation (E=mc^2). However, you should know as well as anyone who can quote that famous equation that energy is not created from mass, but converted from mass...
Caution: Cape does not enable user to fly. - Batman costume warning label (Rolfe, John & Troob, Peter, Monkey Business (Swinging Through the Wall Street Jungle), 2000)
Originally posted by spikegifted
Being a chemist, I've never figured out where the loss in mass amount from... It's just not my field and even physical chemists don't really bother themselves with nuclear physics.
Back to your point, you are completely correct in quoting the equation (E=mc^2). However, you should know as well as anyone who can quote that famous equation that energy is not created from mass, but converted from mass...
Whoops! Did I say created?
Does that mean that mass is stored energy? Does that mean that light is just energy?
AFAIK, it is not a case of 'matter is stored as energy' or visa versa. In a quantum mechanical world (which is what this is all based on), mass is energy and the reverse is also true. One is simply a particular manifestation of another. While all this is true in quantum scale, what is described in quantum scale doesn't translate to the macro world. (Since I'm not a physicist, I can't begin to tell you why this is not the case, but I think it is due to the unlikelihood of the collapse of the wave functions of macro objects...)Originally posted by Big RICHARD
Does that mean that mass is stored energy? Does that mean that light is just energy?
Your reference to ‘light is just energy’ is completely correct. I hope you’d cover the wave-particle duality of light - it can be described as electro-magnetic radiation (wave) as well as photons (particle). Again, as part of ground rules of quantum mechanics (Heisenberg’s Uncertainty Principle), you can’t accurately measure both the position and momentum of a particular particle - by increasing the accuracy of the measurement of one, you’d diminish the accuracy of the other, for amount of energy you deploy to ‘detect’ the particle will change it’s momentum or position. So if we can’t know both of these properties, how can we know if these particles are where we say they are? Well, that’s another one of the ground rules of quantum mechanics - Schrödinger’s Equation (or more commonly known as a ‘wave function’). The solutions to Schrödinger’s Equation give you the probability of finding a given particle in an area of space… I’m not going to dive any deeper here, for it is quickly approaching areas where I’m way out of my depth.There’re hundreds of very readable books (not text books) you can brush up on these interesting topics.

Caution: Cape does not enable user to fly. - Batman costume warning label (Rolfe, John & Troob, Peter, Monkey Business (Swinging Through the Wall Street Jungle), 2000)
I briefly did all those subjects in a-level physics, I was just testing my knowledge and that of others!Originally posted by spikegifted
AFAIK, it is not a case of 'matter is stored as energy' or visa versa. In a quantum mechanical world (which is what this is all based on), mass is energy and the reverse is also true. One is simply a particular manifestation of another. While all this is true in quantum scale, what is described in quantum scale doesn't translate to the macro world. (Since I'm not a physicist, I can't begin to tell you why this is not the case, but I think it is due to the unlikelihood of the collapse of the wave functions of macro objects...)
Your reference to ‘light is just energy’ is completely correct. I hope you’d cover the wave-particle duality of light - it can be described as electro-magnetic radiation (wave) as well as photons (particle). Again, as part of ground rules of quantum mechanics (Heisenberg’s Uncertainty Principle), you can’t accurately measure both the position and momentum of a particular particle - by increasing the accuracy of the measurement of one, you’d diminish the accuracy of the other, for amount of energy you deploy to ‘detect’ the particle will change it’s momentum or position. So if we can’t know both of these properties, how can we know if these particles are where we say they are? Well, that’s another one of the ground rules of quantum mechanics - Schrödinger’s Equation (or more commonly known as a ‘wave function’). The solutions to Schrödinger’s Equation give you the probability of finding a given particle in an area of space… I’m not going to dive any deeper here, for it is quickly approaching areas where I’m way out of my depth.There’re hundreds of very readable books (not text books) you can brush up on these interesting topics.

Your knowledge of physics is similar to mine! It's been a few years since I did any of this!
http://www.bristol.ac.uk/university/...o4_jul2003.pdf
scroll down to pages 12-13 for something relevant to this debate...
Powered by Marmite and Wet Dog
Light Over Water Photography
sound travels through air (or any mass of molecules) by passing the vibrations from one molecule to the next. the speed of sound in water is faster because the molecules are tightly packed. a sonic boom is caused when all of the vibrating frequencies in the air are piled on top of each other because the aircraft is moving the same speed as the sound vibrations can be transferred between molecules (basically its a sound being generated on top of itself making it loud). air technicaly can make sound when it bangs into things (or itself) creating vibrations which are passed on, but it doesnt magically create sound no.
Thanks for that input Pyle, it now makes perfect sense.Originally Posted by PrivatePyle

To err is human. To really foul things up ... you need a computer.
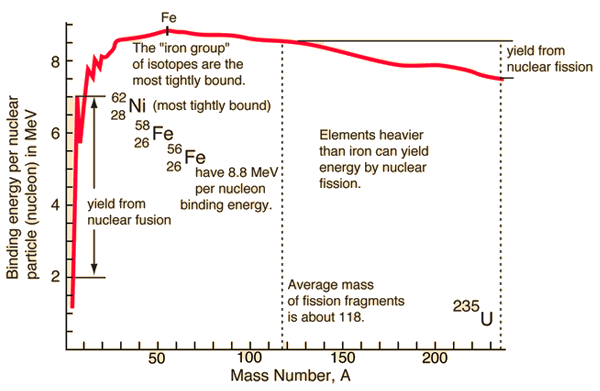
This picture explains it best.
The reason molecules release energy when they undergo fission is because of a quantity called the 'nuclear mass defect'
Essentially this is the difference between the mass of a molecule and the combined masses of all of it's constituent parts(protons, neutrons, electrons)
As can be seen from the curve, as you break up very heavy atoms(Uranium) it gives off smaller atoms which have less of a nuclear mass defect.
Therefore, the extra mass that was holding the Uranium together is released as energy.
As for the original question, shiato storm is correct.
Knight 1: We are now no longer the Knights who say Ni.
Knight 2: NI.
Other Knights: Shh...
Knight 1: We are now the Knights who say..."Ekki-Ekki-Ekki-Ekki-PTANG. Zoom-Boing. Z'nourrwringmm.
as to the original question - any room for a fart joke?
Man what a load of bollocks... im guessing its a slow day at work...
So has the original question turned into can YOU actually here air rather than does air make a sound?
As far as im concerned air makes a sound when its moving everything that moves does (that i can think of anyway) even electrons make noise shot noise white noise etc.
Hehhe brilliant. I am now completely savvy
To err is human. To really foul things up ... you need a computer.
True, Pyle definitely sums it up better
Knight 1: We are now no longer the Knights who say Ni.
Knight 2: NI.
Other Knights: Shh...
Knight 1: We are now the Knights who say..."Ekki-Ekki-Ekki-Ekki-PTANG. Zoom-Boing. Z'nourrwringmm.
There are currently 1 users browsing this thread. (0 members and 1 guests)